Grantee Research Project Results
Final Report: High Information Content Toxicity Screening Using Mouse and Human Stem Cell Models of Endocrine Development and Function
EPA Grant Number: R835163Title: High Information Content Toxicity Screening Using Mouse and Human Stem Cell Models of Endocrine Development and Function
Investigators: Finnell, Richard H.
Institution: Texas A & M University
EPA Project Officer: Aja, Hayley
Project Period: March 1, 2012 through February 28, 2015
Project Amount: $1,200,000
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The world market includes a total of 30,000 industrial chemicals sold at quantities of more than 400 million tons per year. There remains a considerable knowledge gap regarding the relationship between exposure to environmental pollution by industrial chemicals and possible adverse environmental and health effects. This data gap necessitates the implementation of a new vision for toxicity testing and a restructuring of risk assessment procedures. The main objective of our proposed research program is to rigorously demonstrate the reliability and sensitivity of using human and mouse stem cells in high-throughput screening methods designed to provide reliable and highly sensitive risk assessment data for chemicals that impact gamete production, embryonic endocrine development and endocrine signalling. The specific aims of the proposed research program are to characterize developmental and endocrine toxicity of select environmental agents using in vitro toxicity testing of human and mouse stem cells. The data produced in these models will be further developed to produce Bayesian models for developmental toxicity classification and toxicity prediction in humans. Through the incorporation of the chemico-physical properties of the test compound, in vitro toxicity testing using genetically diverse human and mouse stem cell populations, it is possible to ascertain novel data on how these compounds impact on specific intracellular pathways in a dose-response fashion. With our proposed high throughput and high information content screening system, we will contribute to the large federal screening effort to prioritize chemicals for risk assessment. Our contribution will be rapid assessment of chemicals for adverse effects on the development of gametes, adipocytes, and islet β-cells, and adipocyte and β-cell endocrine signaling function in human and murine embryonic stem cells, training and production of hierarchical Bayesian models based on these data, and establishing endpoints that are relevant for human gametic and endocrine development and function.
Toxicity testing in the 21st century must move beyond classical methods of risk assessment. These current methods depend predominately upon animal testing, as described in the guidelines set out by OECD (i.e., repeated-dose and chronic tests, as well as assays using juvenile animals). The cost and time commitments associated with the use of laboratory animals for risk assessment seriously limits efforts for extensive chemical screening. Further, toxicological testing based on the evaluation of predetermined endpoints developed for decades fail to incorporate modern molecular knowledge and methods suggesting that chemicals can disrupt common cell-signaling pathways. Risk assessment is a process that is based on scientific data but is also influenced by extrapolation and uncertainties, the unavoidable consequences of significant existing data gaps in critical steps of the Source-to-Outcome Continuum, which includes chemical fate, transport, metabolism, exposure, dose-response, vulnerability of a given species, and the sensitivity of specific organs at different stages of development.1 The existing regulations of environmental toxicants needs to be revised and the strategies for evaluating chemical for health risks are in need of urgent refinement. The proposed research program makes use of embryonic stem cell screening systems for chemical risk assessment, which will expand the overall throughput, mitigate costs, and recapitulate with fidelity the native developmental and cellular cascades involved in gamete and endocrine development and function that occur in vivo. Such an approach will have highly significant implications for the screening of the increasing number of chemicals released into the environment that have the potential to adversely affect the pre- and post-natal health of hundreds of millions of people in this country, and billions more globally.
Background and Significance: Embryonic stem (ES) cells were isolated during the 1980s from the inner cell mass of developing mouse blastocysts.2,3 When these cells are grown in culture medium supplemented with leukemia inhibitory factor (LIF), or on a layer of embryonic fibroblasts feeder cells, they proliferate undifferentiated and in a pluripotent state. When grown in the absence of feeder cells and LIF, ES cells will differentiate spontaneously. This ability has been exploited for embryonic developmental research, and in making transgenic mice, in that ES cells can be genetically manipulated, and when injected into recipient blastocysts, they will differentiate into various cell types of the organism, including the germ line. Breeding of the resultant chimeric mice thereby allows the creation of a mouse strain carrying highly specific genetic mutations or transgene.
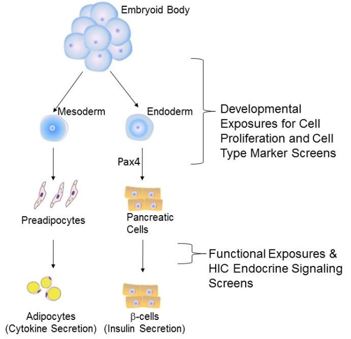
The differentiation of ES cells into spheroid aggregate, which mimics post-implantation embryonic tissues, is termed embryoid body (EB) formation. Cells within developing EBs may subsequently be further differentiated, resulting in the production of committed cell types including: gametes4, adipocytes5 and islet β-cells.6 Future developments in ES cell culture will potentially allow differentiation into any cell type. The combination of these factors enables ES cells to model the effect of developmental toxicants on embryos using a totally in vitro system.
Experimentation on ES cells and EBs has allowed for the in vitro investigation of several aspects of mouse and human development. Due to the difficultly of studying developing mouse and human embryos in vivo, ES cells and EBs have emerged as powerful tools for characterizing the function of embryonic development in vitro. ES cells with transgenic reporter constructs can also be used to detect developmentally regulated genes.7 As proposed in this investigation, ES cell differentiation combined with transgenic expression of reporters provides a powerful tool for rapid observation, isolation and quantitative determination of the gene/protein expression of distinctive cellular markers.
Hypotheses: We hypothesize that high throughput screening (HTS) of genetically diverse human and murine SCs and ES cells can recapitulate observations from in vivo models exposed to endocrine disrupters. Further, in the absence of a priori toxicological data, we hypothesize that HTS of ES cells can be used for classification and prediction of in vivo impact, risk assessment, and prioritizing of chemicals for in vivo testing and epidemiological studies.
Objectives:
- Utilization of mouse and human ESC clones to determine the effect of developmental toxicants and environmental pollutants on ESC differentiation into gametes, adipocytes, and islet β-cells using high throughput methodologies.
- Measure endocrine function and disruption of signaling by environmental pollutants in ESC derived β-cells and adipocytes via measurement of insulin and cytokine production, respectively.
- Utilization of this high throughput in vitro data for the development of risk assessment models that will establish the ability of these data to classify and predict the in vivo effect of environmental pollutants on these endocrine systems.
The proposed research program is expected to: (1) determine the effects of environmental pollutants on gamete, adipogenesis and pancreatic β-cell development; (2) determine the effect of the test compounds on pancreatic endocrine function of insulin production; and (3) provide a rapid and robust tool for risk assessment of chemicals of unknown in vivo toxicity.
Summary/Accomplishments (Outputs/Outcomes):
3.1 Development of HTS and HCS Tools
The overall goal of the proposed research program is to develop toxicity assay tools with which to perform HTS of environmental compounds. We propose to develop an in vitro screening model using mouse and human ESCs as a tool to follow different developmental and differentiation processes. The selected developmental and functional endpoints include:
- Gastrulation (% of each germinal cells type)
- Lipogenesis (% adipose cells)
- Adipose Endocrine Function (Cytokine Profile)
- Pancreatic development (% β-cells)
- Pancreatic Endocrine Function (Insulin Production)
- Gametogenesis (% positive for sperm cell markers)
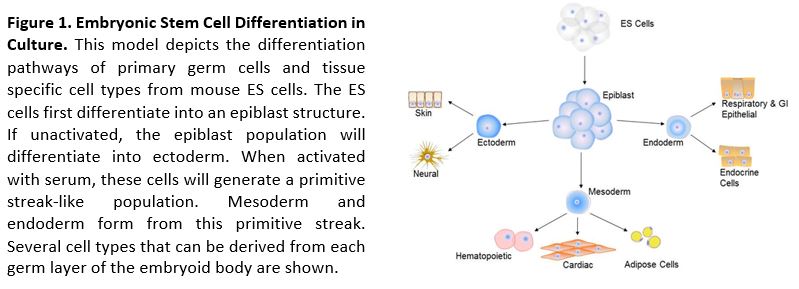
Gastrulation: Immediately post-implantation, a transformation of the cellular structure of the mouse conceptus begins. The implanted blastocyst consists of an inner cell mass (ICM) within the trophectoderm. Over the next 3 days, the polar trophectoderm and the ICM develop into an elongated structure that is made up of the ectoplacental cone connecting the embryo to the uterus, the extraembryonic ectoderm, the epiblast, and a layer of visceral endoderm. Gastrulation begins with the formation of the primitive streak, through which epiblast cells ingress to form the mesoderm and the endoderm. These two tissues and the ectoderm (the descendants of epiblast cells that do not pass through the primitive streak) constitute the three primary germ layers (ectoderm, mesoderm, and endoderm) that contain progenitors of all developmental tissues (reviewed8).
We propose to examine the dynamic consequences of in vivo exposure to environmental compounds on embryonic development via in vitro embryoid body formation. Visualization of the gastrulation phenomenon occurring in vitro faithfully mimics the in vivo process. Furthermore, the establishment of antero-posterior polarity and the formation of a primitive streak-like region, where the cells undergo an epithelial-to-mesenchymal transition (EMT) and differentiate into mesodermal cells in the embryoid body, is dependent on local activation of the Wnt pathway.9 These histological and molecular observations demonstrate the consistency by which the developmental pathways are followed in vitro, as they would be in vivo. Capitalizing on these developmental parallels, the effect of environmental toxicants on the cell fate and germ layer specification of gastrulation can be quickly identified in embryoid bodies derived from ESCs carrying transgenic markers of differentiation and signaling. These unique research reagents enable us to identify the molecular mechanisms being affected by the toxicant in an HTS system. The large quantity of EBs obtained from ESCs, the rapid process of differentiation, and the ever increasing availability of molecular markers with which to follow key processes of differentiation, makes this a highly desirable end point in any HTS testing paradigm.
Antibodies against known markers of germinal and endocrine cell types will be used to quantitate the number of cells that undergo differentiation in the presence of environmental chemicals during gastrulation. The selected antibodies allow the spatial and temporal patterns of definitive classifiers of ectoderm, mesoderm, and endoderm germ layers (Table 1) to be determined. The temporal expression pattern of the cell type marker, spatial distribution and intensity between EB development under normal developmental conditions and following exposure to the test compounds, will be analyzed. By comparing these markers from controls and the environmental pollutants exposed ESCs, the impact of industrial chemicals on the coordinated cellular differentiation events occurring during embryonic development will be determined. The high content information obtained through the exposure to environmental pollutants in conjunction with data existing is expected to produce predictive tools for determining whether environmental pollutants influence the differentiation of cell into primordial germ and endocrine cells by altering the differentiation of early germinal proliferation or cell differentiation.
The above markers will be evaluated in ES cells using immunocytochemistry. All selected antibodies are documented to be confirmed against both mouse and human protein epitopes.
Endocrine Testing: It is well established that pollutants in bodies of water can alter sex differentiation and sexual behavior in aquatic wildlife and birds that feed on exposed fish (review16). These changes are mechanistically driven by endocrine disrupters, but a similar impact on human reproductive health is largely unsupported in population based studies.17 Most of the reported effects of endocrine disrupters in humans are secondary to occupational exposures or pharmaceutical usage. However, tentative links indicate endocrine disrupting pollutants also alter blood glucose homeostasis.18 In model systems, the endocrine disruptor bisphenol-A (BPA) has been shown to exert effects on the endocrine functions of the pancreas, adipocyte differentiation and lipid accumulation.19,20 The endocrine pancreas is critical to hormone production and glucose homeostasis, which parallels the exocrine activities for digestion. Further, these effects of BPA on adipocytes and the pancreas can be influenced by in utero exposures.20,21 This proposal uses ESC in vitro differentiation to generate adipocytes and endoderm-like pancreatic precursor cells, as indicated by transcriptome analysis (Finnell Laboratory, unpublished data). The adipocytes are PPAR γ positive, and the continued differentiation of these pancreatic cells results in the formation of C-peptide- and insulin-positive cells. Pancreatic development can be further influenced by Pax4, which activates ES cell–derived pancreatic differentiation into islet-like clusters, with insulin-secretory granules. These endocrine cells can recapitulate glucose homeostasis when transplanted into streptozotocin-treated diabetic mice.6 Gene expression analysis and immunocytochemistry analysis of ESCs-derived, embryonic, and adult pancreas indicates ESC-derived endocrine cells mirror embryonic stages of development.22 Therefore, we propose to utilize this system to determine the in vitro impact of endocrine disrupters and predict the in utero risk of environmental chemical exposures on pancreatic and adipocyte development and function.
4.1 Technical Approach
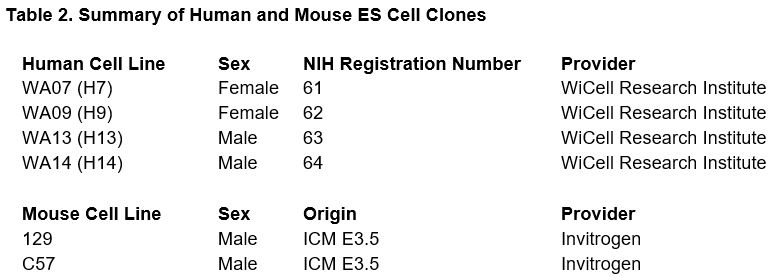
The techniques utilized in our proposed research program are in daily usage in the Finnell Laboratory, and will be primarily based on our standard ES cell expansion protocol. This protocol allows for expansion of the ES cell clones from single vials to high capacity 96-wells, 24-wells, 60mm, or 100mm plates. All expansions utilize standard M15 media and Soriano ES Feeder cells (SNL 76/7 STO). This procedure produces stock replacement, quality control aliquots, and several experimental stocks. While a variety of differentiation techniques can be utilized to produce specific cell types from ES cells, the ES cells in these studies (Table 2) will be plated into 96-well plates and directed to differentiate into embryoid bodies.
These mouse ES cells are commercially available (Invitrogen) and the human ES cells have been previously described23 and approved to comply with Executive Order (EO) 13505, entitled Removing Barriers to Responsible Scientific Research Involving Human Stem Cells.
Development of the HTS assays: There are three basic methods that are utilized for the culture of embryoid bodies: liquid suspension culture in bacterial-grade dishes, culture in methylcellulose semisolid media, and culture in hanging drops. The use of commercially available 96-well plates (Thermo Fisher Scientific, Rochester, NY) coated with 2-methacryloyloxyethyl phosphorylcholine (MPC), prevents ES cells from adhering to the plastic, and makes it possible to perform microscopic observations of embryoid body in culture.24 These procedures are already developed for a parallel study as part of a USEPA funded STAR research program. Each human clone is plated horizontally across two rows, 24-wells total per clone, such that untreated wells from each cell line may be used to analyze the response of treatment for inter and intra-cell line responses. Dose responses are determined by 10-fold dilutions across the columns of the plate in triplicate. A single pollutant may be assayed per plate producing five dose response points, four untreated-control wells for each of the four human ES cell lines. This same scheme is also utilized for the mouse cell lines, with the exception that two cell lines are used and therefore two test environmental pollutants/compounds may be assayed per 96-well plate.
A laser scanning confocal imager (In Cell Analyzer 3000, GE Healthcare) capable of live cell imaging (controlled at 35-37°C, 70% humidity, 5% CO2) in 96 and 384 well plate formats and providing dynamic, continuous autofocus at subcellular (0.6 μm max) resolution will be used for HIC and HTS imaging. The expression of the cell type specific markers for control and environmental pollutant-exposed ES cells, EBs, and differentiated endocrine cells will be used to monitor and observe those coordinated cellular differentiation and functional events that are altered by endocrine disrupters. Time lapse images will be taken of EBs (phototoxicity will also be determined, and image acquisition frequency will be no more than one image per 5 minutes or 30% the rate and duration required for phototoxicity) to address the effect of toxicants on embryonic growth and differentiation. A comparison of cell population numbers, expression markers of differentiation, via FACs sorting (Guava EasyCyte 8HT, Millipore) will also be made to precisely quantify the differentiation effects caused by the test compound.
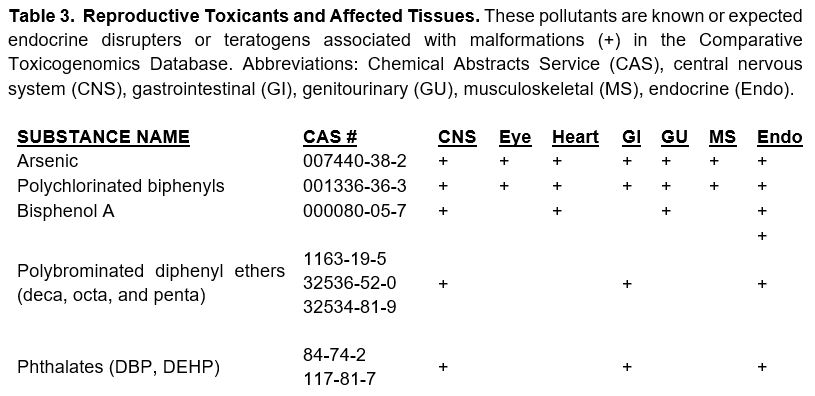
Selection of Test Compounds: The current list (circa 2007) of Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA) chemicals was used to query the Comparative Toxicogenomics Database (CTD).25 This list was reviewed and queried to produce a list of real-world environmental pollutants that function as endocrine disrupters, (Table 3).26 This data was then compiled and used. These compounds will be used as training compounds for in vitro testing (Table 3).
Embryonic Stem Cell Culture: The human and mouse cells are maintained on SNL76/7 fibroblast feeder cells that express that expresses LIF and grown as a monolayers in defined medium of Knockout DMEMM (Invitrogen) supplemented with serum replacement factor (Invitrogen), 1% non-essential amino acids (Invitrogen), 2 mM L-glutamine (Invitrogen) and 0.1 mM β-mercapto-ethanol (Sigma-Aldrich). The cells are maintained in a humidified atmosphere at 37°C and 5% CO2.
Embryoid Bodies for High-Throughput Differentiation and Testing: Embryoid bodies are obtained via 96-well culture in complete medium without LIF.27 In brief, stem cell suspensions (1.2·106 cells/well) are placed in AggreWell 400 plates (Stem Cell Technologies) and incubated in a humidified atmosphere at 37°C and 5% CO2. After 24-hours of AggreWell culture, uniformly sized EBs (1,000 cells per EB) are formed and these were subsequently plated, 10 per well, on ultra-low attachment 96-well culture plates (Cole Parmer).
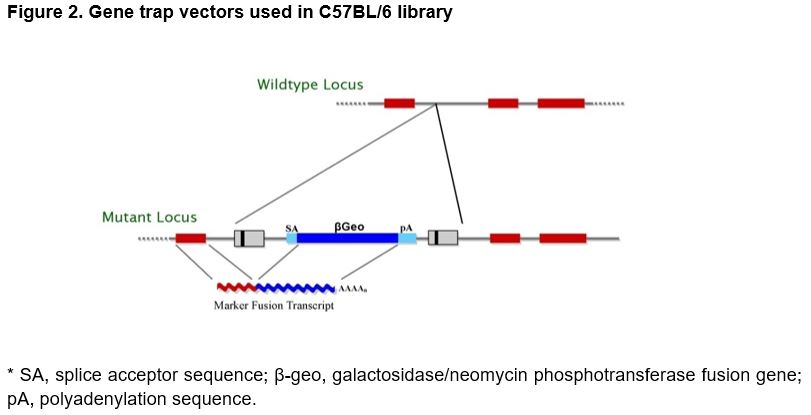
Mouse Embryonic Stem Cell Library for Gamete Selection: The ES cell clone library for gamete production in mice utilizes gene traps for gene inactivation.28 In this approach, a promoterless marker/reporter gene (β-geo, a functional fusion between the β-galactosidase and neomycin resistance genes) is introduced into ESCs. Selection for expression of the gene requires transcription from a cellular promoter. Consequently, a mutation in a cellular gene and the activity of the tagged gene can be followed by staining for β-galactosidase activity.
Vector Design: The Texas Institute for Genomic Medicine (TIGM) ES cell clones were generated using several different retroviral gene trap vectors that all contain the 5' selectable marker β-geo for identification of successful gene trap events.28 The β-geo marker also allows for in vivo expression studies of the trapped gene through in situ hybridization, immunohistochemistry, or other β-galactosidase-based assays (Figure 2).
Gamete Production from ESCs: ESC differentiation methods combined with transgenic expression of a reporter provides a powerful tool for rapid observation and quantitative determination of the gene/protein expression of distinctive cell-type specific markers. We propose to use these technologies to measure endocrine disrupter impact on the expression of gamete cells produced from
Differentiation of ESCs into Somatic Endocrine Cell Types: Differentiation of mouse ESCs into pancreatic endocrine (insulin-producing) cells requires spontaneous differentiation of the EBs into progenitor cells of ecto-, meso- and endodermal lineages followed by culture in secondary differentiation media, i.e., DMEM with N-2 additives (Invitrogen) to produce pancreatic lineage progenitors or DMEM with retinoic acid (Sigma) to produce adipocytes.5,6
Testing Compounds for Impact on Developmental Tissues: ES cell differentiation cultures are exposed from the EB stage at day 3 onwards to test chemicals (Table 3) or vehicle (0.20% DMSO). All chemicals are serially diluted to determine a median infective dose (ID50), i.e., the concentration at which 50% of EBs do not undergo proper differentiation compared to the controls, determined by cell type specific transgenic marker expression and/or cell type behavior (e.g., 50% decrease in the number of adipocytes). We propose to determine the spatial distribution and intensity of expression of cell-type markers throughout differentiation in 96-well high-throughput plates. This period will encompass gastrulation, endocrine, adipocyte, or gamete development, while the high-throughput format will allow the plating of experimental and control replicates in addition to the culture of multiple ES cell clones in a single experiment. By comparing the number of cells positive for cellular markers for each of these processes from control and environmental pollutant-exposed embryos, we propose to monitor and observe those coordinated cellular differentiation events that are altered by endocrine disrupting chemicals. Images will be taken daily of the embryoid bodies to address the effect of toxins on embryonic growth and differentiation. A comparison of cell population numbers and expression of markers of differentiation, via high throughput confocal microscopy and flow cytometry (In Cell Analyzer 3000 and Guava EasyCyte 8HT, respectively) will be used to precisely quantitate the differentiation effects caused by the chemical pollutant.
Data Gathered using HIC Screening Kits:
Apoptosis 1 Multiparameter Kit—Multiplexed assay quantifies three features of apoptosis: nuclear morphology (condensation/ fragmentation), mitochondrial mass/potential, and cytoskeletal changes (f-actin content) specifically and reliably in whole cells.
Cy Quant Cell Proliferation Kit—fluorescence enhancement upon binding to cellular nucleic acids. The fluorescence emission of the dye-nucleic acid complexes correlates linearly with cell number over a large range using a wide variety of cell types.
Cytokine and Insulin Screen—fully quantitative microELISA-based test where sixteen distinct capture antibodies have been absorbed to each well of a 96-well plate in a defined array (Quansys Biosciences).
The initial screen of four human and two mouse cell lines (Table 2) and eight test chemicals (Table 3), 48 combinations, will initially be tested across 24 wells in a dose response study, using replicates of four with five serial dilutions and one negative control.
Dose response and IC50 will be determined and are provided by two serial (1/10) concentrations above and below the median observed or expected in blood/serum reported in the literature for exposed populations. For chemicals with sharp slopes, dilutions will be adjusted based upon expected biometric curves. All data will be analyzed and tested for multidimensional patterns using hierarchical clustering (Hierarchical Clustering Explorer 3.0, University of Maryland). Additionally, predictive models of development and function will be generated by our co-investigator Dr. Ken Drake of Seralogix, Austin, TX. These models are based upon the chemical structures and properties, as well as the collective data across all chemicals cell lines, dose response concentrations, and time points tested.
We have extensive experience in testing analyte concentrations (e.g. folate) and immune responses (e.g., IgG) using the microELISA system described above.41,42
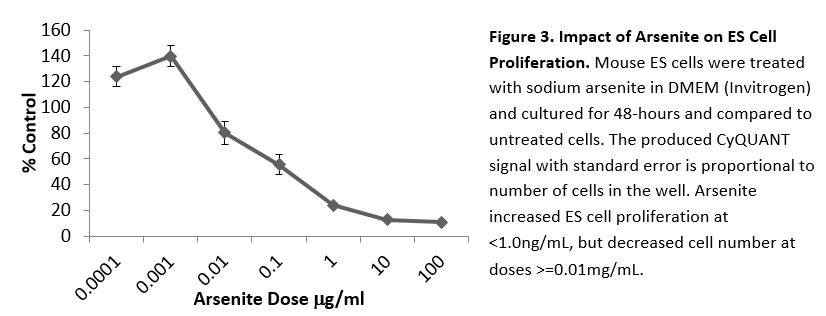
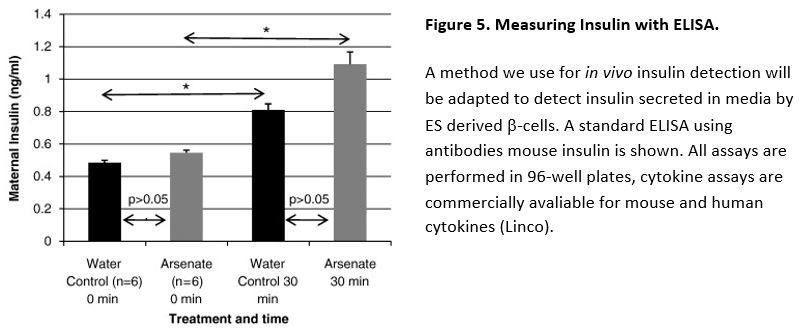
In a previously published study of the impact of arsenate on insulin and glucose signaling, we determined that insulin levels were slightly higher 30 min after a glucose challenge in the arsenate-treated dams versus controls, but this difference was not statistical significant.43
Predictive Biological Systems Modeling
Modeling has the potential to elucidate how chemicals interact with biological targets that disrupts important pathways, resulting in an adverse outcome. The heterogeneous toxicity datasets to be collected herein, will be employed in a Bayesian network modeling approach. Bayesian learning is a mature machine learning method that can be used to classify chemicals and employed to predict biological responses representing toxicity. An existing systems biology Bayesian framework (Seralogix, LLC, BioSignature Analysis Framework, www.seralogix.com) will be leveraged to create an ensemble of hiearchical Bayesian models, similar to those employed by Langdon44 and enhanced to create models that are trained with chemical structural fingerprints45 and substructure toxicophores46 and toxicity response datasets for prediction of biological process and pathway disruption as inferred from end-point measurements (described previously). The Bayesian method will allow models to be created that identify and classify biological responses by species (mouse and human ES cell strain) and genetic diversity (male and female human ES cells). A strength of this framework is that it can create models that include both discrete and continous varaibles and include prior toxicity and chemical structure information from public databases (EPA ECOTOX, etc.). The model performance will be assessed by traditional methods of sensitivity and specificity testing. The Seralogix modeling framework was selected for this effort because of its extensibility to include higher dimensional “omic“ data and pathway network structure learning at the systems biology level. This framework was developed with the support of National Insitutes of Health funding, and has capabilities for data management and computational pipeline to accomodate the data generation and processing demands of high throughput testing. The framework also produces web-based reports that will made available to the scientific community via the internet after appropriate institutional vetting and approvals. These reports will provide a comprehesive profile of each chemical cell line test, with associated statistical and modeling results.
5.1 Expected Results and Benefits
This research program is expected to provide predictive information on the in vivo effect of environmental pollutants. Specifically, the effects of chemical pollutants will be tested on ES derived cells that model gastrulation, spermatogenesis, and development and endocrine function of adipocytes and pancreatic cells. These approaches will thereby allow risk assessment modeling with data, which represents critical developmental processes and tissues. The disruption of any of these processes would be expected to produce a variety of metabolic disorders. While this investigation specifically focuses on the in vitro platform for testing environmental toxicants in a high-throughput format, the Finnell Laboratory has the experience and expertise to validate chemicals for toxicology and teratogenicity in vivo. This is demonstrated by the results of data on both pharmaceutical (e.g., valproic acid) and environmental teratogens (e.g., arsenic) tested in our various mouse models.47,48
The platform utilized in this investigation will provide direct benefit to the general public because it will provide a much needed tool for evidence-based risk assessment predictions on the endocrine activity and toxicity of environmental pollutants. This screen will allow identification of pollutants that pose the greatest potential for causing endocrine health problems through environmental exposures. Our specific focus on a wide variety of developing tissues tested in high-throughput formats also provides for replication and culture variations that are expected to minimize both the false-negative and false-positive results. This will have the effect of saving both time and money, in that impact on the endocrine system can be ranked by consistent effect (biological deviation from control values) on the precise tissue expected to be affected, prior to any costly downstream in vivo, epidemiological or environmental work being conducted.
6.1 Innovation
The proposed research program involves the testing of gamete production from embryonic stem cells of mouse and human origin. As important as the demonstration of the utility of this HIC/HTS is to the field of environmental toxicology and public health, it has a very significant ancillary or spinoff technology that will have a very high impact on the field of transgenic knockout mouse production. While the methods are similar to those used in our laboratory for neural differentiation, defining a set of cellular marker to select for functional gametes is critical. While testing these gametes for mouse production or for clinical applications in human infertility treatments is beyond the scope of this application, these data are expected to outline and establish foundational knowledge and intellectual property in the area. The methods may thereby establish the ability to rapidly create genetically modified mouse models for the global mouse research community.
The ability to collect, rapidly analyze, and create predictive models from the high throughput toxicity datasets is an important innovation in that the results can be quickly reviewed and interpreted by research scientists and policy makers. Such reviews can be used to modify and guide future experiments, as well as to inform regulatory bodies. The Seralogix modeling platform can already incorporate genomic, proteomic, and metabolomics data from other high throughput measurement platforms. Such data integration, coupled with the high throughput toxicity testing technique (which systematically perturbs the biological system) can be used to learn and identify the complex interactions of chemical toxicants disrupting biology pathways (visionary plans).
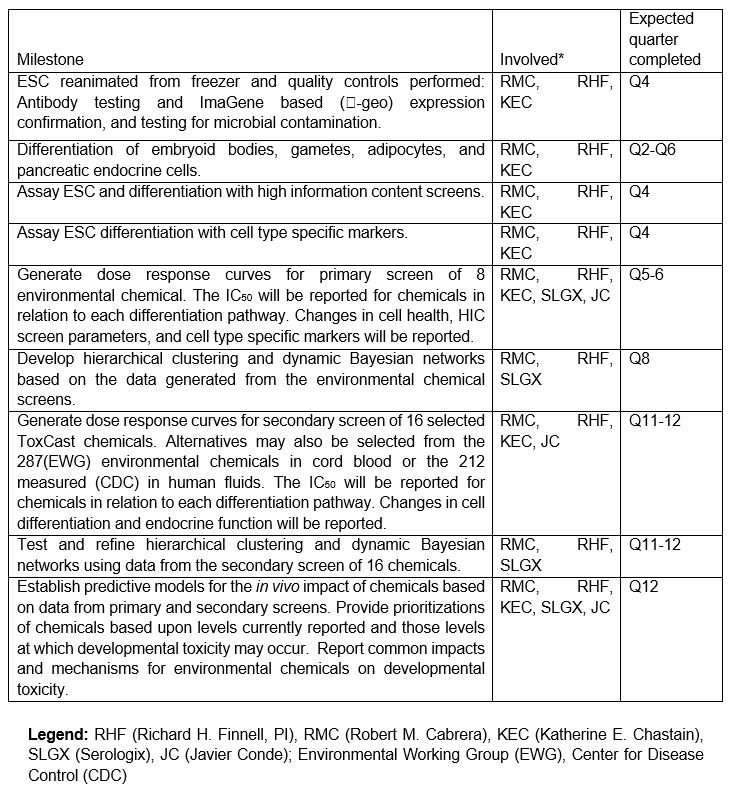
7.1 Expected TimeLine
8.1 Results
Adipocytes
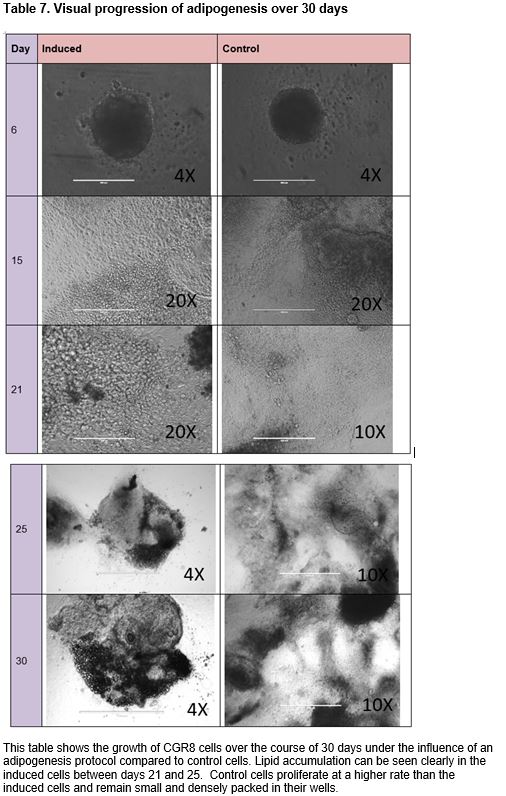
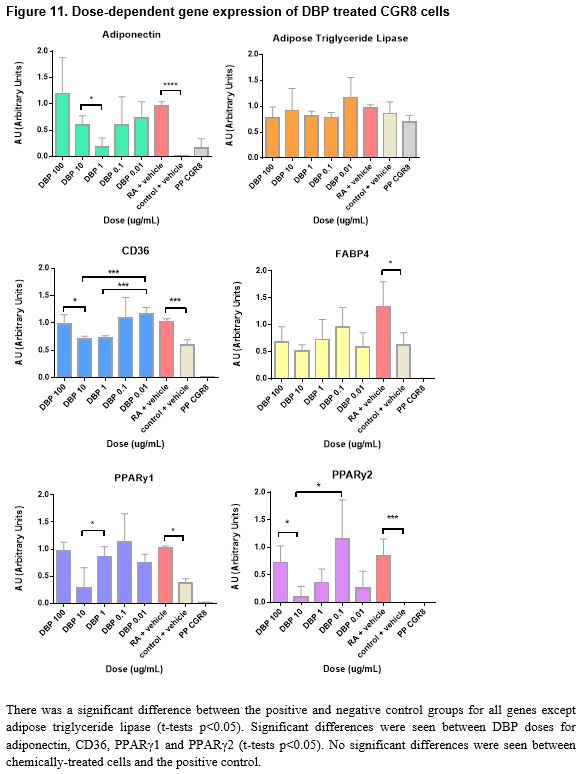
The prevalence of obesity, diabetes, and related disorders has increased at a rate that is indicative of environmental influence in addition to food intake and genetic predisposition. Endocrine disrupting chemicals (EDCs) found in commonly used household products such as plastics and metal cookware have demonstrated an exponential increase in prevalence in the past decade that is parallel to the increase in obesity rates. The binding of EDCs to nuclear hormone receptors can influence hormone-mediated pathways and affect downstream target genes. The differentiation of pluripotent stem cells to mature adipocytes in the process of adipogenesis involves coordination of a network of transcriptional factors and is greatly determined by a variety of signaling molecules. Exposure to EDCs during an early ontogenetic window of differentiation may influence cellular commitment to an adipogenic fate. This study examines the effect of three endocrine disrupting chemicals, mono-(2-ethylhexyl) phthalate (DEHP), dibutyl phthalate (DBP), and pentadecafluorooctanoic acid (PDFO/PFOA) on the adipogenic outcome of CGR8 mouse embryonic stem cells when treated with varying dosages that mimic environmental exposure during an early window of differentiation. The capacity of these chemicals to promote adipogenesis was assessed by quantifying changes in adipogenic marker gene expression (adiponectin, adipose triglyceride lipase/patatin-like phospholipase domain containing 2 (PNPLA2), cluster of differentiation 36/FAT (fatty acid translocase) (CD36), fatty acid binding protein 4 (FABP4), peroxisome proliferator-activated receptor gamma 1 (PPARγ1), and peroxisome proliferator-activated receptor gamma 2 (PPARγ2)) in comparison to untreated cells using qPCR. Though an increased level of lipid accumulation and adipogenic growth was seen in vitro, and significant differences were found between concentrations of chemicals, no significant differences in gene expression were found between chemically treated cells and untreated groups.
During the past few decades, the production and usage of synthetic organic and inorganic chemicals has increased exponentially. Interestingly, this increase in synthetic chemical production precedes and runs parallel to the exponential increase of obesity rates.49 The environmental obesogen hypothesis proposes that this correlation is not coincidental, and environmental pollutants may be a contributing factor to the obesity epidemic.50 Epidemiological studies have shown that there is a connection between the development of obesity and the production of various synthetic chemicals.51 Endocrine disrupting chemicals found in many plastics, pesticides, and additives affect hormone-mediated pathways by binding to and influencing nuclear hormone receptors.50 Differentiation of cells and other cell-mediated processes that occur during fetal development are greatly determined and influenced by a variety of signaling molecules.52
Adipogenesis is the process of cell differentiation during which precursor preadipocytes become adipocytes under the increased expression of adipocytic genes such as fatty acid binding protein (FABP4) and peroxisome proliferator-activated receptor-gamma (PPARγ).53 Adipogenic differentiation involves several distinct ontogenetic windows: undifferentiated growth, induction, and terminal differentiation.51 The differentiation of embryonic stem cells to mature adipocytes involves coordination of a complex network of transcriptional factors and signaling intermediates from a variety of pathways.54 Early exposure to endocrine disrupting chemicals may influence cellular differentiation and affect the likelihood of embryonic stem cells becoming mature adipocytes. Ultimately, the overall question pertains to whether maternal exposure during early pregnancy can lead to fetal predisposition for obesity and diabetes.
Perfluoroalkyl acids (PFAAs) are a class of synthetic chemicals that have hydrophobic and oleophobic properties that make them ideal surfactants. They are used in a wide variety of commercial and industrial applications, including stain resistant sprays, nonstick cooking surfaces, and food contact paper coatings.55 Pentadecafluorooctanoic acid (PDFO/PFOA) is a type of PFAA used as a polymerization aid in the manufacture of fluoropolymers, often found in metal cookware products.56 Prenatal exposure to low doses of PFOA in mice has been shown to increase non-neoplastic liver lesions, and result in compromised postnatal survival as well as sex-specific alterations in pubertal maturation.57,58
To date, most studies have focused on the impact of endocrine disrupting chemicals on adipogenesis using 3T3-L1 preadipocytes, mesenchymal stem cells, or CGR8 embryonic stem cells during later windows of differentiation. In order to explore the adipogenic impact of DBP, DEHP, and PDFO on early embryonic commitment and differentiation. The current study involved exposing mouse CGR8 embryonic stem cells to the endocrine disrupting chemicals during early induction at various concentrations that mimic environmental exposure. Cells exposed to EDCs were predicted to have a higher level of adipogenic activity and adipogenic gene expression.
Materials and Methods
All studies were performed as independent duplicate experiments. DBP, DEHP, and PDFO were obtained from Sigma-Aldrich (Taufkirchen, Germany) and diluted in methanol for addition to media during in vitro culture studies. The concentrations of each chemical selected for use in this study were based roughly on exposure levels present in the environment as ascertained by the Environmental Protection Agency (EPA).
Cell Culture
The murine embryonic stem cell (ESC) line CGR8 was seeded (4x1000 cells/cm²) using 0.25% trypsin or trypsin/EDTA; 5% CO2; 37°C , and cultured on mitomycin-treated STO mouse embryonic fibroblasts to prevent differentiation. Sub-confluent cultures (70%-80%) were split if not used immediately. The cells were fed CGR8 Media (Glasgow's MEM (GMEM), 10% fetal bovine serum (FBS), Penicillin Streptomycin 100X, GLUTamax 100X, non-essential amino acids 100X, 2-Mercaptoethanol 2000X)) and medium was changed daily. Confluent cultures were harvested from the STO feeder cells using TrypLE Express (Life Technologies, India); to harvest the cells, the STO + ESC 20 mm culture dish was washed with Dulbecco’s phosphate-buffered saline (DPBS) three times and treated with 1mL TrypLE for three minutes. The enzyme was deactivated with 10mL GMEM and the entire volume of the plate was transferred to a new non-gelatinized 20 mm plate. The plate was incubated for 30 minutes, during which time the STO feeder cells should adhere to the bottom of the plate while the CGR8 cells remain suspended in solution. The cells were collected along with the media and transferred to a new non-gelatinized plate for another 30 minutes and the process was repeated for a total of three non-gelatinized 20 mm plates and three transfers with a total incubation time of 1.5 hours. After the third transfer, the cells were counted with a hemocytometer and seeded 2000 cells per well in a round-bottom, 4% pluronic-acid treated, ultra-low adherence 96-well plate with 200 uL of CGR8 media per well for embryoid body (EB) formation.
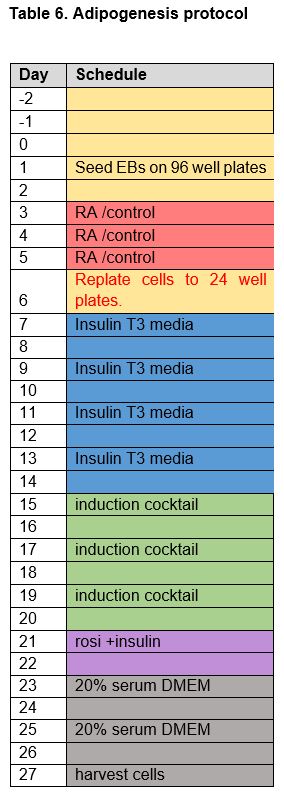
Adipogenesis Protocol and Chemical Treatment
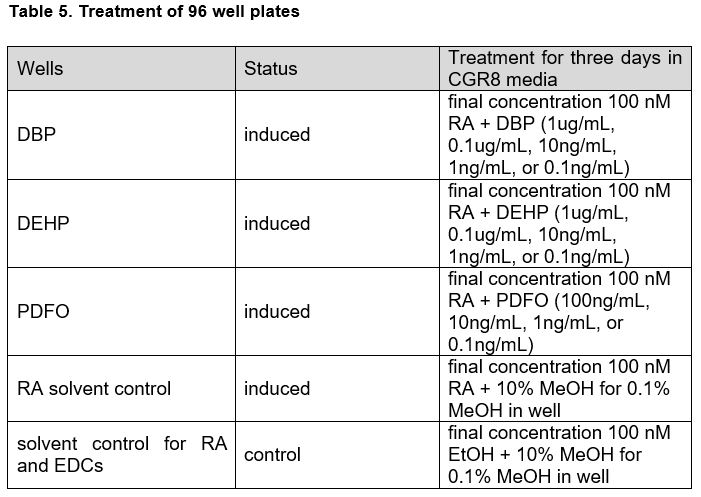
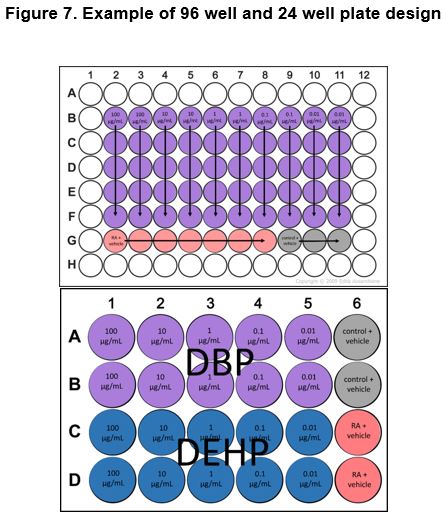
The cells were left to grow for one day as an embryoid body, after which wells were subjected to a first induction with retinoic acid (1mM working stock) appropriate chemicals in CGR8 media for three days. Concentrations of retinoic acid, chemicals, and vehicle were doubled for the first day of induction to account for dilution by pre-existing media in the wells. Experimental wells were treated with a final concentration of 100 nM retinoic acid (RA) + EDC in various concentrations: DBP and DEHP wells were treated with final concentrations of 1 µg/mL, 0.1 µg/mL, 10 ng/mL, 1 ng/mL, or 0.1 ng/mL using 100X working stocks while PDFO wells were treated with final concentrations of 100 ng/mL, 10 ng/mL, 1 ng/mL, or 0.1 ng/mL with 100X working stocks. To account for the methanol the EDCs were dissolved in, retinoic acid solvent control wells were treated with RA + 10% MeOH. To account for both the methanol solvent for the EDCs and the ethanol solvent of the retinoic acid, solvent control wells for RA and EDCs were treated with EtOH + 10% MeOH.
After 3 days of exposure to retinoic and EDCs, the cells were replated to CytoOne gelatinized 24-well plates. Induced wells were seeded 5 EBs per well and control wells were seeded 3 EBs as the control EBs proliferate faster than treated EBs. After replating to 24-well plates, the cells are left to grow for one day before starting insulin/triiodothyronine (T3) treatment. Insulin/T3 treatment for eight days, changing the media every other day, allows expansion of adipocytes from embryoid bodies.59 T3 is shown to potentiate insulin signaling and increase insulin sensitivity (Lin, 2011). Induced cells were treated with final concentrations of 100 nM insulin and 2 nM T3 in 20% FBS STO media (DMEM (Knockout Dulbecco's Modified Eagle's Medium), 20% FBS, Penicillin Streptomycin 100X, GLUTamax 100X, non-essential amino acids (NEAA) 100X). Control cells were treated with 10% FBS STO media (DMEM (Knockout Dulbecco's Modified Eagle's Medium, 10% FBS, Penicillin Streptomycin 100X, GLUTamax 100X, non-essential amino acids (NEAA) 100X). Following the eight days of ins/T3 treatment, the experimental wells were subjected to a second induction consisting of stock insulin, 20 mM rosiglitazone, 1mM dexamethasone, 1M 3-isobutyl-1-methylxanthine (IBMX), and 333 mM indomethacin, and medium was changed every other day. The control wells were treated with 0.1% DMSO (solvent control). A total of six days of exposure to the second induction cocktail is followed by an additional two days of only stock insulin and 20 mM rosiglitazone to further enhance adipogenesis.60 The cells were subsequently incubated in either 20% FBS DMEM (induced cells) or 10% FBS DMEM (control cells) for 5 days, changing the media every other day. Counting from the day cells were seeded on pluronic-acid treated 96-well plates, cultures were harvested in 500 uL Trizol Reagent after washing with DPBS on the 26th day of adipogenic treatment and stored at -80°C.
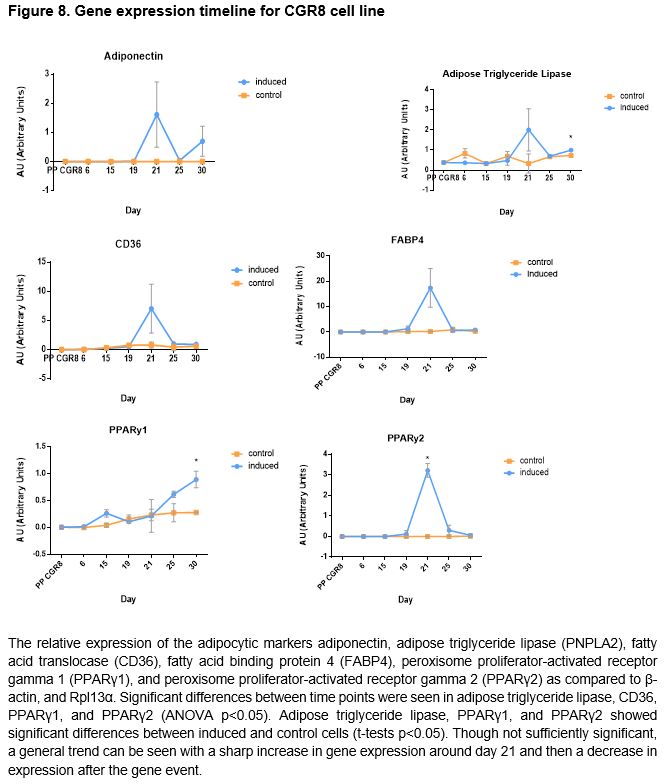
Gene Expression Timeline of CGR8 Stem Cells
To ascertain an opportune time point to harvest cells for gene expression analysis with qPCR, a timeline of gene expression of selected genes was created. Cells were grown as dictated by the aforementioned adipogenesis protocol excluding the addition of endocrine disrupting chemicals during retinoic acid treatment. Cells were harvested from wells in 500 uL of Trizol Reagent after washing with DPBS on days 6, 15, 19, 21, 25, and 30 and stored at -80°C.
Real time polymerase chain reaction
Total RNA extraction was performed on cell samples in Trizol using the miRNeasy Micro Kit from Qiagen, and cDNA synthesis using RNA samples was performed using the QuantiTect Reverse Transcription Kit from Qiagen. Quantitative real-time PCR was performed with primer sets for adipocyte markers adiponectin, adipose triglyceride lipase/patatin-like phospholipase domain containing 2 (PNPLA2), cluster of differentiation 36/FAT (fatty acid translocase) (CD36), fatty acid binding protein 4 (FABP4), peroxisome proliferator-activated receptor gamma 1 (PPARγ1), and peroxisome proliferator-activated receptor gamma 2 (PPARγ2). mRNA levels were normalized with β-actin, and Rpl13α levels and presented as a relative fold change to control.
Results
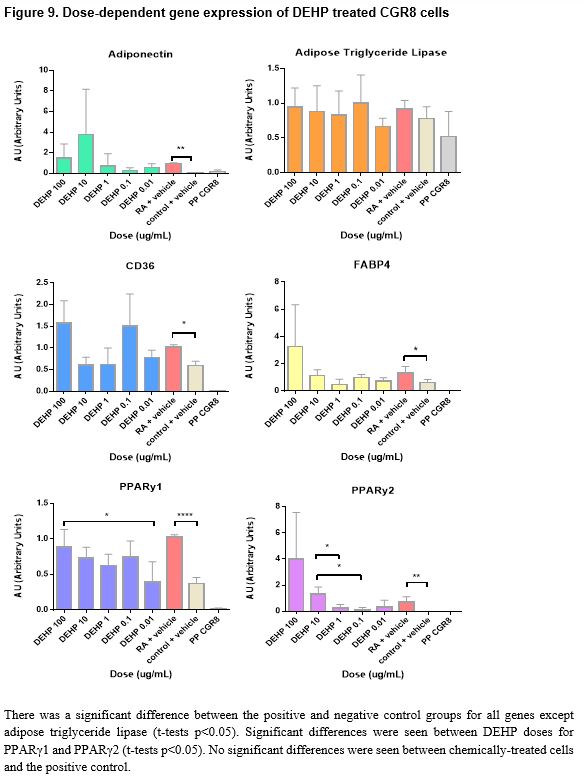
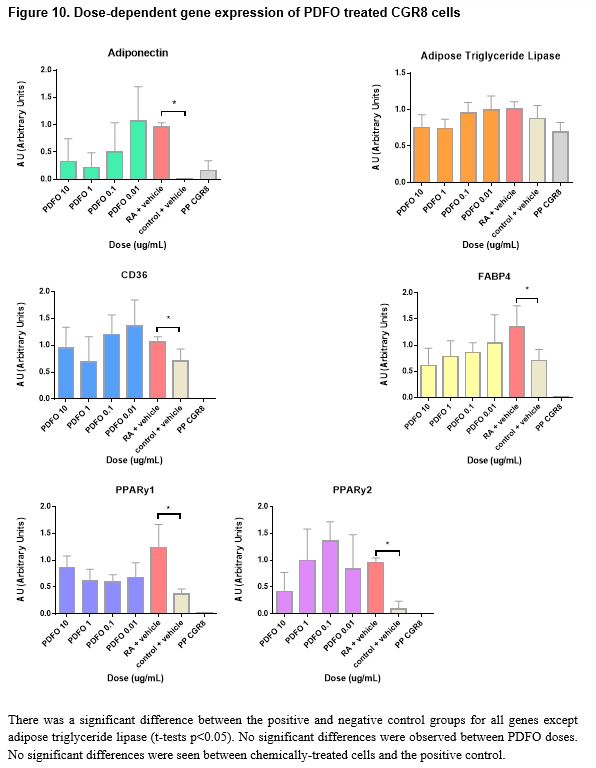
Pluripotent CGR8 embryonic stem cells differentiated in an adipocytic direction after treatment. Gene expression events seem to occur after day 19; though not sufficiently significant in all genes but PPARγ2, an increase of expression at day 21 followed by a decrease in expression by day 25 is seen in all genes except for PPARγ1, which increases after day 21. No significant results were found between chemical-treated cells (DEHP, PDFO, and DBP) and positive controls; however, differences between doses in DEHP and DBP-treated cells can be seen (p < 0.05).
Conclusions
The disruption of nuclear hormone receptors by EDCs has been seen to modulate sex steroid and thyroid hormone actions leading to changes in sexual development and cell growth.61 EDC exposure can lead to altered development and homeostatic control over adipogenesis and energy balance; when treated with several EDCs including bisphenol A and dicyclohexyl phthalate, 3T3-L1 pre-adipocytes were found to show increased lipid accumulation and upregulated expression of adipocytic proteins.62 Although there are several studies that have examined the effects of EDCs on pre-adipocytes and mesenchymal stem cells, the implications of environmental endocrine disruptors on the development of adipose cells from pluripotent embryonic stem cells when treated at an early ontogenetic window have been insufficiently investigated. This study evaluated the effects of three different endocrine disrupting chemicals, bis(2-ethylhexyl) phthalate (DEHP), dibutyl phthalate (DBP), and pentadecafluorooctanoic acid (PDFO) on the differentiation of pluripotent CGR8 mouse embryonic stem cells into adipocytes, treated during an early window of differentiation. Embryoid bodies were treated with the EDCs at varying concentrations during the first induction with retinoic acid. With minimal time to conduct a large number of experiments, the experiment to ascertain a timeline of gene expression was conducted in parallel to the chemical experiments in order to determine an optimal window of time to harvest the cells for gene expression quantification. From the timeline data, a change in gene expression seems to occur after day 19; a sharp increase at day 21 followed by a sharp decrease in expression at day 25 is seen for all genes excluding PPARγ1, which continues to increase past day 25.
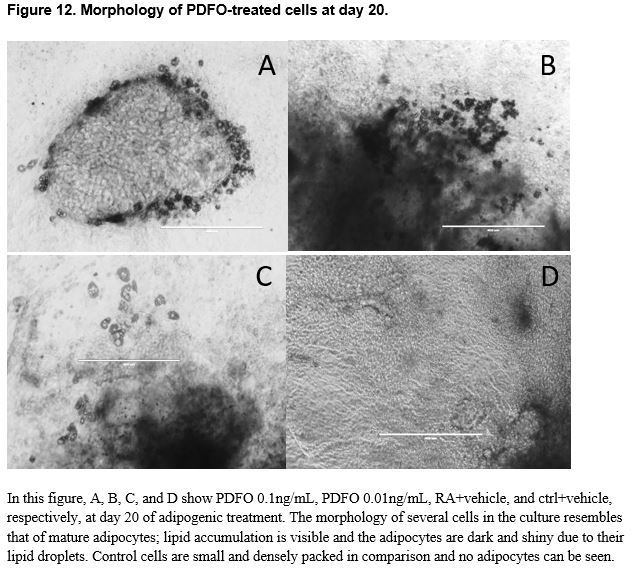
The chemically-treated cells were harvested on either day 26 or day 27 when the cells were observed to be mature adipocytes, presumably past the optimal window of gene expression as indicated by the timeline data. Morphologically, the cells at day 26 resembled mature adipocytes more than the cells at day 21; it appears that the gene expression event for terminal adipocyte differentiation precedes any morphological events that may occur at a later time. Visually, in vitro, of the three chemicals tested in this study, PDFO at a concentration of 0.1ng/mL and 0.01ng/mL resulted in the largest number of adipocytes. However, analysis of the gene expression data obtained from the PDFO experiments showed no significant results for PDFO both within different concentrations and as compared to the positive control, which followed the same adipogenic protocol with no additional chemicals. The results for PDFO show increased expression at 0.1 ng/mL and 0.01 ng/mL, in accordance with what was seen in vitro, but the experimental data was insufficient for statistically supporting any visual differences seen in the cell culture plate. DEHP-treated cells also showed no increase in adipogenic gene expression as compared to the positive control, though there were differences between different concentrations of DEHP: significant differences were seen between DEHP doses for PPARγ1 and PPARγ2 (t-tests p < 0.05). PPARγ is an adipocyte-specific gene that commits the cell to an adipogenic fate, and the binding of EDCs on this receptor can influence the activity of PPARγ and direct the cell to an adipose lineage.63 Although not sufficiently significant, from the figures, it appears that with increased concentration of DEHP, there is increased adipocytic gene expression aside from the higher expression observed at DEHP 0.1ng/mL. Additional replicates of the experiment would potentially provide more conclusive data as the values obtained were close to being, but not, significant. DBP at a concentration of 100 ng/mL and 0.01 ng/mL showed the highest increase in gene expression, and several genes, namely adiponectin, CD36, and the PPARy genes, showed significant differences in expression between DBP concentrations, but no DBP-treated groups were found to significantly upregulate the chosen adipocytic marker genes relative to the positive control. All genes aside from adipose triglyceride lipase showed higher expression in the positive controls for all experiments as compared to the negative controls. This finding is indicative to successful differentiation of the pluripotent stem cells into mature adipocytes under the adipogenesis protocol used in this study.
In conclusion, based on accumulated data, DEHP and DBP affect adipogenesis in a dose-dependent manner; however, treatment of CGR8 stem cells with the chemicals alongside retinoic acid during the first induction did not show increased adipocyte marker gene expression as compared to the positive control. Further investigation is required to examine the gene expression levels of treated cells when harvested on day 21, where, according to the timeline of gene expression, a change in expression seems to occur. Additional replicates of the experiments conducted in this study would provide more conclusive data as many concentrations were close to being significant and having a triplicate of duplicates would offer stronger statistical analyses. Degradation of RNA or cDNA could have resulted in results that didn’t accurately portray gene events in cell samples. Loss of adipocytes, which tend to be buoyant, and other cells during harvesting and transfer between containers could have skewed gene expression representation of the true cell population in samples. Investigation into the mechanism of interaction between the chemicals and retinoic acid to induce adipogenic differentiation would be an interesting supplement to current data. Although the chemicals used in this study are known to disrupt reproduction and promote adipogenesis when treated at a later window of differentiation, the experiments conducted in this study failed to prove its effects on early adipogenesis in CGR8 stem cells. DEHP, PDFO, and DBP are only three out of a large number of endocrine disrupting chemicals that humans are exposed to throughout their lifetimes. Examination of possible effects from other endocrine disrupting chemicals outside of the three used in this study would provide further insight into the possible correlation between synthetic chemical exposure and increased incidences of obesity.
Pancreatic Signaling
Glucose uptake using a fluorescent glucose analog is an established technique to provide information on the impact of small molecules with respect to glucose uptake. The glucose analog 2-NBDG is a fluorescently-labeled deoxyglucose analog that is used to directly monitor glucose uptake by living cells and tissues. Specifically, we performed the present study to assess the uptake of glucose (2-NBDG) induced by skin (MEFs) and muscle (C2C12) cells. Additionally, we determined the impact of insulin signaling on the glucose transporter (GLUT4) using live-cell imaging and fluorescently-labeled transporter (GLUT4-GFP).
Materials and Methods
Cytotoxicity
Biological Material: Live Cells
Cellular background: MEF cells, mES cells
Fluorescent Ligand: 2-PI
Glucose Uptake
Biological Material: Live Cells
Cellular background: C2C12 cells, MEF cells
Fluorescent Ligand: 2-NBDG
Glucose Transporter Activity
Biological Material: Live Cells
Cellular background: C2C12 cells
Fluorescent Tracer: GLUT4-GFP
Cellular assays were performed using mouse embryonic fibroblasts (MEFs) and myoblasts (ATCC). Cell proliferation rates were determined using automated cell counting of cells after 48hr exposure in complete medium (DMEM w/ 10% FBS). Cell are then washed thrice with 1X PBS and incubated with Accutase (Invitrogen) for 10 min at 37°C, neutralized with 10% FBS in 1XPBS, and analyzed by flow cytometry. Cell toxicity was determined by adding propidium iodide (PI) to suspended cells prior to flow cytometry. The PI is a red-fluorescent DNA stain that is excluded by live-healthy cells but will enter dead cells in a population.
Glucose uptake assays were carried out in a final incubation volume of 50 µl consisting of 2-NBDG (100 µM) in 1XPBS w/ Ca+, Mg+ (PBS++). The same buffer is used for washing cells. Insulin was used as a positive control. Cells are culture to 70-90% confluence and then washed twice with PBS++. Cells are incubated for 30 minutes at 37°C in PBS++. Nuclear stain is added, HCS NuclearMask stain (blue, Invitrogen) and testing compounds are added (insulin or vehicle). All testing compounds are prepared in 10%DMSO and diluted 1:100 for a final concentration of 0.1% DMSO (vehicle). Cells are incubated for 30min at 37°C. Cells are washed twice with PBS++ and incubated with 2-NBDG for 10 minutes. Cells are washed thrice with PBS++ and then imaged for microscopy in PBS++. Alternatively, the final PBS wash is removed and cells are incubated with Accutase (Invitrogen) for 10 min at 37°C, neutralized with 10% FBS in 1XPBS, and analyzed by flow cytometry.
The localization of glucose transporter (GLUT4-GFP) was performed in final incubation volume of 0.505 mL, consisting of 500 µl of Hank’s Buffer (Invitrogen) and 5 µl of tested compounds. Insulin was used as positive controls and vehicle (0.1% DMSO) as the negative control.
Cells were rinsed in Hank’s Buffer thrice and then incubated in Hank’s at 37°C for 1 hour. Test compounds are added (insulin, or vehicle). All test compounds are prepared in 10% DMSO and diluted 1:100 for a final concentration of 0.1% DMSO (vehicle), and the cells were imaged using laser-confocal microscopy (Leica TCS SP5 X) with FITC fluorescent filters for changes in GLUT4-GFP localization. Time-lapse images (TIF) were collected and post-processed using Image-J (NIH) for video construction.
All test compounds of interest were initially dissolved in 100% DMSO. The saturated solution was diluted 1:10 in 1XPBS; this 100X stock solution is 400 µM. The working concentrations (1X) were 4µM, followed by 1:10 dilutions into 0.1% DMSO. The resulting six dilutions ranged from 4 µM to 0.04nM in triplicate. Dose curves were analyzed using either Harmony software on the Operetta (Perkin Elmer) or Excel (Microsoft) for flow cytometry, which was performed on the BD Accuri (BD Biosciences).
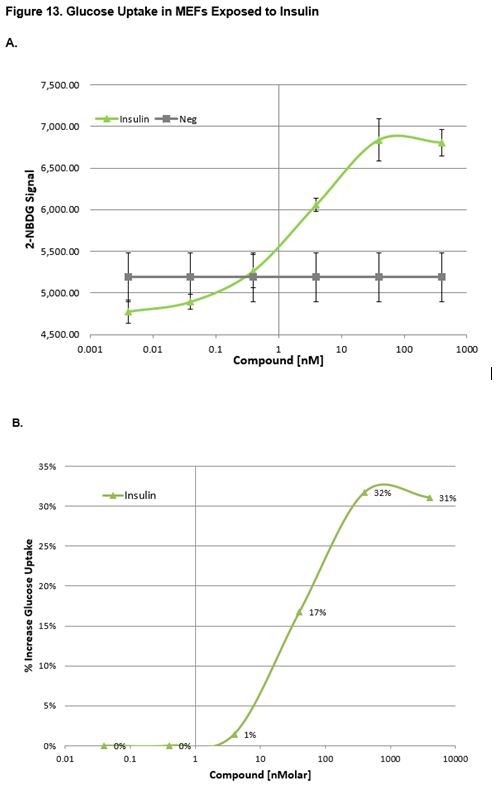
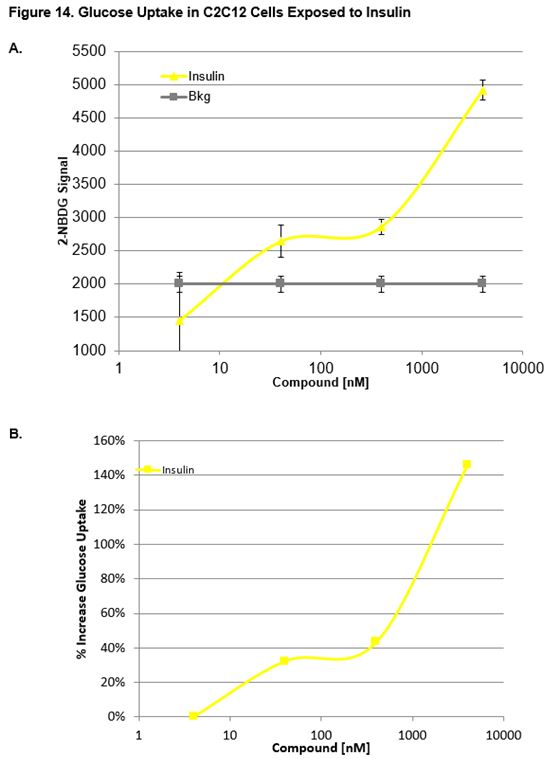
Effect of Insulin on MEFs and C2C12 Cells
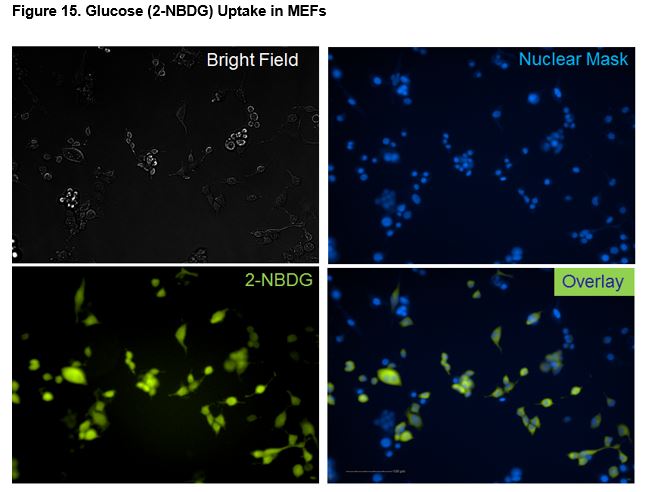
Figs. 13a, 13b and 14a, 14b show the uptake curves for test compounds. Figures 13a and 14a indicate the fluorescent signal of glucose (2-NBDG) taken up by the cell. Figures 13b and 14b indicate the relative glucose uptake as a percent change relative to vehicle treated (0.1% DMSO) cells. As expected, insulin produced a significant increase in glucose uptake relative to the vehicle. Figure 15 shows images taken of MEFs after incubation with Insuling. Cells were stained with Nuclear Cell Mask (blue, FITC) and glucose (green, FITC, 2-NBDG) uptake was imaged on the Operetta HCS (Perkin Elmer).
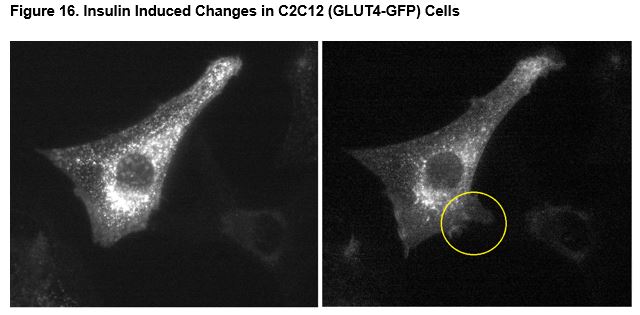
Effect of Insulin on GLUT4 in C2C12 Cells
Figure 16 show changes in cells due to insulin and indicates that the fluorescent signal (GLUT4-GFP) is predominately located around the nucleus prior to the insulin exposure. Exposure to insulin (4000 nM) produced a decrease in nuclear GLUT4-GFP and rapid ruffling of filopodia at the surface of the cell (Figure 16, right panel). There was no indication of changes in nuclear localization of GLUT4-GFP or ruffling of filopodia in any time-lapse imaging of untreated or vehicle treated cells (supplemental data available).
Conclusions
Myoblast cells transfected with GLUT4-GFP demonstrated decreased nuclear localization of GLUT4-GFP after simulation of insulin (4000 nM). Exposure to insulin induced rapid ruffling of the cell membrane and numerous projections (filopodia) began billowing out of the cell. The projections appear to be directly related to an increase in cycling of GLUT4 to and from the cellular surface and cellular uptake of glucose by GLUT4. These assays may be used for screening high-throughput screening for compounds for disruption or activation of insulin-dependent glucose uptake.
Conclusions:
Initial attempts were made in order to produce gametes from mouse embryonic stem cells. These studies proved beyond the time and outside the scope of endocrine development and function. We did establish human embryonic stem cell lines H7, H9, H13, and H14. Each of these lines were obtained from WiCell and expanded on Matrigel using previously established protocols. All cell lines were also sent out for cytogenetic analysis by WiCell, reports available. These cells were utilized for pancreatic beta cell differentiation, as previously described.64 A considerable amount of time and resources were spent establishing the protocols for producing pancreatic beta cells, and the problem in beta cell production was traced to the insulin-transferrin-selenium-fibronectin supplement. We produced our own medium supplement for differentiation, and for reasons that we were unable to determine, this medium resulted in cellular toxicity. We ultimately switched to a commercial source (Sigma-Aldrich) of the supplement and were able to obtain insulin producing beta cells. Gene expression of pancreatic genes was confirmed and insulin production was determined and confirmed by ELISA. Our initial experiments indicated that endocrine disrupting chemicals were not specific modifiers of pancreatic beta-cell development. In retrospect, this is supported by the low incidence of neonatal diabetes mellitus, estimated at 1 in 500,000.65 In support of this, clinical studies also indicate that decreased insulin secretion in the fetus or newborn appears as a genetic disease, with mutations in genes encoding proteins that alter the function of the pancreatic beta-cell.66
In summary, we have shown the application of adipocyte differentiation as a screen for environmental chemical testing. We analyzed markers for cellular differentiation in response to endocrine disrupting toxicants, and demonstrated the impact of developmental toxicants on biomarker expression, specifically adipokines. We have also demonstrated that some endocrine disruptors are potent morphogens, or able to modify the activity of retinoic acid, while others are potent activators of adipokines. These activities are expected to have detrimental impacts on embryonic and/or fetal endocrine, specifically adipocyte, development.
References:
- Kavlock, R. and D. Dix, Computational toxicology as implemented by the U.S. EPA: providing high throughput decision support tools for screening and assessing chemical exposure, hazard and risk. J Toxicol Environ Health B Crit Rev, 2010;13(2-4):197-217.
- Martin, G.R., Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A, 1981;78(12):7634-8.
- Evans, M.J. and M.H. Kaufman, Establishment in culture of pluripotential cells from mouse embryos. Nature, 1981;292(5819):154-6.
- Geijsen, N., et al., Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature, 2004;427(6970):148-54.
- Dani, C., et al., Differentiation of embryonic stem cells into adipocytes in vitro. J Cell Sci, 1997;110( Pt 11):1279-85.
- Schroeder, I.S., et al., Differentiation of mouse embryonic stem cells to insulin-producing cells. Nat Protoc, 2006;1(2):495-507.
- Gossler, A., et al., Mouse embryonic stem cells and reporter constructs to detect developmentally regulated genes. Science, 1989;244(4903):463-5.
- Tam, P.P. and D.A. Loebel, Gene function in mouse embryogenesis: get set for gastrulation. Nat Rev Genet, 2007;8(5):368-81.
- ten Berge, D., et al., Wnt signaling mediates self-organization and axis formation in embryoid bodies. Cell Stem Cell, 2008;3(5):508-18.
- Dziadek, M. and E. Adamson, Localization and synthesis of alphafoetoprotein in post-implantation mouse embryos. J Embryol Exp Morphol, 1978;43:289-313.
- Arceci, R.J., et al., Mouse GATA-4: a retinoic acid-inducible GATA-binding transcription factor expressed in endodermally derived tissues and heart. Mol Cell Biol, 1993;13(4):2235-46.
- Wilkinson, D.G., S. Bhatt, and B.G. Herrmann, Expression pattern of the mouse T gene and its role in mesoderm formation. Nature, 1990;343(6259):657-9.
- Hopwood, N.D., A. Pluck, and J.B. Gurdon, MyoD expression in the forming somites is an early response to mesoderm induction in Xenopus embryos. EMBO J, 1989;8(11):3409-17.
- Bouwens, L., et al., Cytokeratins as markers of ductal cell differentiation and islet neogenesis in the neonatal rat pancreas. Diabetes, 1994;43(11):1279-83.
- Chawla, A., et al., Peroxisome proliferator-activated receptor (PPAR) gamma: adipose-predominant expression and induction early in adipocyte differentiation. Endocrinology, 1994;135(2):798-800.
- Vos, J.G., et al., Health effects of endocrine-disrupting chemicals on wildlife, with special reference to the European situation. Crit Rev Toxicol, 2000;30(1):71-133.
- Safe, S., Clinical correlates of environmental endocrine disruptors. Trends Endocrinol Metab, 2005;16(4):139-44.
- Ropero, A.B., et al., Bisphenol-A disruption of the endocrine pancreas and blood glucose homeostasis. Int J Androl, 2008;31(2):194-200.
- Alonso-Magdalena, P., et al., The estrogenic effect of bisphenol A disrupts pancreatic beta-cell function in vivo and induces insulin resistance. Environ Health Perspect, 2006;114(1):106-12.
- Rubin, B.S. and A.M. Soto, Bisphenol A: Perinatal exposure and body weight. Mol Cell Endocrinol, 2009;304(1-2):55-62.
- Cederroth, C.R. and S. Nef, Fetal programming of adult glucose homeostasis in mice. PLoS One, 2009;4(9):e7281.
- Rolletschek, A., et al., Characterization of mouse embryonic stem cell differentiation into the pancreatic lineage in vitro by transcriptional profiling, quantitative RT-PCR and immunocytochemistry. Int J Dev Biol, 2010;54(1):41-54.
- Thomson, J.A., et al., Embryonic stem cell lines derived from human blastocysts. Science, 1998;282(5391):1145-7.
- Koike, M., H. Kurosawa, and Y. Amano, A Round-bottom 96-well Polystyrene Plate Coated with 2-methacryloyloxyethyl Phosphorylcholine as an Effective Tool for Embryoid Body Formation. Cytotechnology, 2005;47(1-3):3-10.
- Davis, A.P., et al., Comparative Toxicogenomics Database: a knowledgebase and discovery tool for chemical-gene-disease networks. Nucleic Acids Res, 2009;37(Database issue):D786-92.
- Lewis, R.J., N.I. Sax, and Knovel (Firm), Sax's Dangerous Properties of Industrial Materials. 10th ed. 2003, New York: John Wiley & Sons.
- De Smedt, A., et al., Optimisation of the cell cultivation methods in the embryonic stem cell test results in an increased differentiation potential of the cells into strong beating myocard cells. Toxicol In Vitro, 2008;22(7):1789-96.
- Hansen, G.M., et al., Large-scale gene trapping in C57BL/6N mouse embryonic stem cells. Genome Res, 2008;18(10):1670-9.
- Kerkis, A., et al., In vitro differentiation of male mouse embryonic stem cells into both presumptive sperm cells and oocytes. Cloning Stem Cells, 2007;9(4):535-48.
- Eguizabal, C., et al., Generation of primordial germ cells from pluripotent stem cells. Differentiation, 2009;78(2-3):116-23.
- Nayernia, K., et al., In vitro-differentiated embryonic stem cells give rise to male gametes that can generate offspring mice. Dev Cell, 2006;11(1):125-32.
- Yu, Z., et al., Dazl promotes germ cell differentiation from embryonic stem cells. J Mol Cell Biol, 2009;1(2):93-103.
- Nayernia, K., et al., Stem cell based therapeutical approach of male infertility by teratocarcinoma derived germ cells. Hum Mol Genet, 2004;13(14):1451-60.
- Toyooka, Y., et al., Embryonic stem cells can form germ cells in vitro. Proc Natl Acad Sci U S A, 2003;100(20):11457-62.
- Jin, J., et al., Catsper3 and Catsper4 are essential for sperm hyperactivated motility and male fertility in the mouse. Biol Reprod, 2007;77(1):37-44.
- Richardson, R.T., et al., Nuclear autoantigenic sperm protein (NASP), a linker histone chaperone that is required for cell proliferation. J Biol Chem, 2006;281(30):21526-34.
- Tarnasky, H., et al., Gene trap mutation of murine outer dense fiber protein-2 gene can result in sperm tail abnormalities in mice with high percentage chimaerism. BMC Dev Biol, 2010;10:67.
- Sapiro, R., et al., Male infertility, impaired sperm motility, and hydrocephalus in mice deficient in sperm-associated antigen 6. Mol Cell Biol, 2002;22(17):6298-305.
- Iwanaga, A., et al., Ablation of the scaffold protein JLP causes reduced fertility in male mice. Transgenic Res, 2008;17(6):1045-58.
- Pires-daSilva, A., et al., Mice deficient for spermatid perinuclear RNA-binding protein show neurologic, spermatogenic, and sperm morphological abnormalities. Dev Biol, 2001;233(2):319-28.
- Cabrera, R.M., et al., Autoantibodies to folate receptor during pregnancy and neural tube defect risk. J Reprod Immunol, 2008l;79(1):85-92.
- Salojin, K.V., et al., A mouse model of hereditary folate malabsorption: deletion of the PCFT gene leads to systemic folate deficiency. Blood, 2011;117(18):4895-904.
- Hill, D.S., et al., Arsenate-induced maternal glucose intolerance and neural tube defects in a mouse model. Toxicol Appl Pharmacol, 2009;239(1):29-36.
- Langdon, S.R., et al., Predicting cytotoxicity from heterogeneous data sources with Bayesian learning. J Cheminform, 2010;2(1):11.
- Guha, R. and S.C. Schurer, Utilizing high throughput screening data for predictive toxicology models: protocols and application to MLSCN assays. J Comput Aided Mol Des, 2008;22(6-7):367-84.
- Williams, D.P., Toxicophores: investigations in drug safety. Toxicology, 2006;226(1):1-11.
- Wlodarczyk, B.J., et al., Arsenic-induced gene expression changes in the neural tube of folate transport defective mouse embryos. Neurotoxicology, 2006;27(4):547-57.
- Finnell, R.H., et al., Common hierarchies of susceptibility to the induction of neural tube defects in mouse embryos by valproic acid and its 4-propyl-4-pentenoic acid metabolite. Teratology, 1988;38:313-320.
- Baillie-Hamilton, P.F., Chemical toxins: a hypothesis to explain the global obesity epidemic. J Altern Complement Med, 2002;8(2):185-92.
- Decherf, S. and B.A. Demeneix, The obesogen hypothesis: a shift of focus from the periphery to the hypothalamus. J Toxicol Environ Health B Crit Rev, 2011;14(5-7):423-48.
- Biemann, R., et al., Endocrine disrupting chemicals affect the adipogenic differentiation of mesenchymal stem cells in distinct ontogenetic windows. Biochem Biophys Res Commun, 2012;417(2):747-52.
- Gicquel, C. and Y. Le Bouc, Hormonal regulation of fetal growth. Horm Res, 2006;65 Suppl 3:28-33.
- Gregoire, F.M., C.M. Smas, and H.S. Sul, Understanding adipocyte differentiation. Physiol Rev, 1998;78(3):783-809.
- Rosen, E.D. and O.A. MacDougald, Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol, 2006;7(12):885-96.
- Watkins, D.J., et al., Exposure to perfluoroalkyl acids and markers of kidney function among children and adolescents living near a chemical plant. Environ Health Perspect, 2013;121(5):625-30.
- Powley, C.R., et al., Determination of perfluorooctanoic acid (PFOA) extractable from the surface of commercial cookware under simulated cooking conditions by LC/MS/MS. Analyst, 2005;130(9):1299-302.
- Filgo, A.J., et al., Perfluorooctanoic Acid (PFOA)-induced Liver Lesions in Two Strains of Mice Following Developmental Exposures: PPARalpha Is Not Required. Toxicol Pathol, 2015;43(4):558-68.
- Lau, C., et al., Effects of perfluorooctanoic acid exposure during pregnancy in the mouse. Toxicol Sci, 2006;90(2):510-8.
- Kansara, M.S., et al., Physiological concentrations of insulin and T3 stimulate 3T3-L1 adipocyte acyl-CoA synthetase gene transcription. Am J Physiol, 1996;270(5 Pt 1):E873-81.
- McTernan, P.G., et al., Insulin and rosiglitazone regulation of lipolysis and lipogenesis in human adipose tissue in vitro. Diabetes, 2002;51(5):1493-8.
- Hofmann, P.J., L. Schomburg, and J. Kohrle, Interference of endocrine disrupters with thyroid hormone receptor-dependent transactivation. Toxicol Sci, 2009;110(1):125-37.
- Sargis, R.M., et al., Environmental endocrine disruptors promote adipogenesis in the 3T3-L1 cell line through glucocorticoid receptor activation. Obesity (Silver Spring), 2010;18(7):1283-8.
- Janesick, A. and B. Blumberg, Minireview: PPARgamma as the target of obesogens. J Steroid Biochem Mol Biol, 2011;127(1-2):4-8.
- Segev, H., et al., Differentiation of human embryonic stem cells into insulin-producing clusters. Stem Cells, 2004;22(3):265-74.
- von Muhlendahl, K.E. and H. Herkenhoff, Long-term course of neonatal diabetes. N Engl J Med, 1995;333(11):704-8.
- Stoy, J., et al., Clinical and molecular genetics of neonatal diabetes due to mutations in the insulin gene. Rev Endocr Metab Disord, 2010;11(3):205-15.
Journal Articles on this Report : 34 Displayed | Download in RIS Format
| Other project views: | All 44 publications | 34 publications in selected types | All 34 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Agopian AJ, Bhalla AD, Boerwinkle E, Finnell RH, Grove ML, Hixson JE, Shimmin LC, Sewda A, Stuart C, Zhong Y, Zhu H, Mitchell LE. Exon sequencing of PAX3 and T (brachyury) in cases with spina bifida. Birth Defects Research Part A: Clinical and Molecular Teratology 2013;97(9):597-601. |
R835163 (Final) |
Exit |
|
|
Cabrera RM, Finnell RH, Zhu H, Shaw GM, Wlodarczyk BJ. Transcriptional analyses of two mouse models of spina bifida. Birth Defects Research Part A: Clinical and Molecular Teratology 2012;94(10):782-789. |
R835163 (Final) |
Exit |
|
|
Denny KJ, Coulthard LG, Jeanes A, Lisgo S, Simmons DG, Callaway LK, Wlodarczyk B, Finnell RH, Woodruff TM, Taylor SM. C5a receptor signaling prevents folate deficiency-induced neural tube defects in mice. Journal of Immunnology 2013;190(7):3493-3499. |
R835163 (Final) |
Exit Exit |
|
|
Denny KJ, Coulthard LG, Finnell RH, Callaway LK, Taylor SM, Woodruff TM. Elevated complement factor C5a in maternal and umbilical cord plasma in preeclampsia. Journal of Reproductive Immunology 2013;97(2):211-216. |
R835163 (Final) |
Exit Exit |
|
|
Denny KJ, Jeanes A, Fathe K, Finnell RH, Taylor SM, Woodruff TM. Neural tube defects, folate, and immune modulation. Birth Defects Research Part A: Clinical and Molecular Teratology 2013;97(9):602-609. |
R835163 (Final) |
Exit |
|
|
Denny KJ, Kelly CF, Kumar V, Witham KL, Cabrera RM, Finnell RH, Taylor SM, Jeanes A, Woodruff TM. Autoantibodies against homocysteinylated protein in a mouse model of folate deficiency-induced neural tube defects. Birth Defects Research Part A: Clinical and Molecular Teratology 2016;106(3):201-207. |
R835163 (Final) |
Exit |
|
|
Farkas SA, Bottiger AK, Isaksson HS, Finnell RH, Ren A, Nilsson TK. Epigenetic alterations in folate transport genes in placental tissue from fetuses with neural tube defects and in leukocytes from subjects with hyperhomocysteinemia. Epigenetics 2013;8(3):303-316. |
R835163 (Final) |
Exit Exit Exit |
|
|
Fathe K, Palacios A, Finnell RH. Brief report novel mechanism for valproate-induced teratogenicity. Birth Defects Research Part A: Clinical and Molecular Teratology 2014;100(8):592-597. |
R835163 (Final) |
Exit |
|
|
Gulvady AA, Murphy EJ, Ciolino HP, Cabrera RM, Jolly CA. Glycerol-3-phosphate acyltransferase-1 gene ablation results in altered thymocyte lipid content and reduces thymic T cell production in mice. Lipids 2013;48(1):3-12. |
R835163 (Final) |
Exit |
|
|
Gulvady AA, Ciolino HP, Cabrera RM, Jolly CA. Resveratrol inhibits the deleterious effects of diet-induced obesity on thymic function. The Journal of Nutritional Biochemistry 2013;24(9):1625-1633. |
R835163 (Final) |
Exit |
|
|
Hansler A, Chen Q, Gray JD, Ross ME, Finnell RH, Gross SS. Untargeted metabolite profiling of murine embryos to reveal metabolic perturbations associated with neural tube closure defects. Birth Defects Research Part A: Clinical and Molecular Teratology 2014;100(8):623-632. |
R835163 (Final) |
Exit |
|
|
Hen N, Shekh-Ahmad T, Yagen B, McDonough JH, Finnell RH, Wlodarczyk B, Bialer M. Stereoselective pharmacodynamic and pharmacokinetic analysis of sec-Butylpropylacetamide (SPD), a new CNS-active derivative of valproic acid with unique activity against status epilepticus. Journal of Medicinal Chemistry 2013;56(16):6467-6477. |
R835163 (Final) |
Exit Exit Exit |
|
|
Kur E, Mecklenburg N, Cabrera RM, Willnow TE, Hammes A. LRP2 mediates folate uptake in the developing neural tube. Journal of Cell Science 2014;127(Pt 10):2261-2268. |
R835163 (Final) |
Exit Exit |
|
|
Lai S-C, Nakayama Y, Sequeira JM, Wlodarczyk BJ, Cabrera RM, Finnell RH, Bottiglieri T, Quadros EV. The transcobalamin receptor knockout mouse: a model for vitamin B12 deficiency in the central nervous system. FASEB Journal 2013;27(6):2468-2475. |
R835163 (Final) |
Exit |
|
|
Lei Y, Zhu H, Duhon C, Yang W, Ross ME, Shaw GM, Finnell RH. Mutations in planar cell polarity gene SCRIB are associated with spina bifida. PLoS One 2013;8(7):e69262 (8 pp.). |
R835163 (Final) |
Exit Exit |
|
|
Lei Y, Zhu H, Yang W, Ross ME, Shaw GM, Finnell RH. Identification of novel CELSR1 mutations in spina bifida. PLoS One 2014;9(3):e92207 (8 pp.). |
R835163 (Final) |
Exit Exit |
|
|
Lupo PJ, Canfield MA, Chapa C, Lu W, Agopian AJ, Mitchell LE, Shaw GM, Waller DK, Olshan AF, Finnell RH, Zhu H. Diabetes and obesity-related genes and the risk of neural tube defects in the National Birth Defects Prevention Study. American Journal of Epidemiology 2012;176(12):1101-1109. |
R835163 (Final) |
Exit Exit |
|
|
Lupo PJ, Chapa C, Nousome D, Duhon C, Canfield MA, Shaw GM, Finnell RH, Zhu H; National Birth Defects Prevention Study. A GCH1 haplotype and risk of neural tube defects in the National Birth Defects Prevention Study. Molecular Genetics and Metabolism 2012;107(3):592-595. |
R835163 (Final) |
Exit Exit |
|
|
Lupo PJ, Mitchell LE, Canfield MA, Shaw GM, Olshan AF, Finnell RH, Zhu H, National Birth Defects Prevention Study. Maternal-fetal metabolic gene-gene interactions and risk of neural tube defects. Molecular Genetics and Metabolism 2014;111(1):46-51. |
R835163 (Final) |
Exit |
|
|
Martinez-Una M, Varela-Rey M, Cano A, Fernandez-Ares L, Beraza N, Aurrekoetxea I, Martinez-Arranz I, Garcia-Rodriguez JL, Buque X, Mestre D, Luka Z, Wagner C, Alonso C, Finnell RH, Lu SC, Martinez-Chantar ML, Aspichueta P, Mato JM. Excess S-adenosylmethionine reroutes phosphatidylethanolamine towards phosphatidylcholine and triglyceride synthesis. Hepatology 2013;58(4):1296-1305. |
R835163 (Final) |
Exit Exit Exit |
|
|
Martinez-Una M, Varela-Rey M, Mestre D, Fernandez-Ares L, Fresnedo O, Fernandez-Ramos D, Gutierrez-de Juan V, Martin-Guerrero I, Garcia-Orad A, Luka Z, Wagner C, Lu SC, Garcia-Monzon C, Finnell RH, Aurrekoetxea I, Buque X, Martinez-Chantar ML, Mato JM, Aspichueta P. S-Adenosylmethionine increases circulating very-low density lipoprotein clearance in non-alcoholic fatty liver disease. Journal of Hepatology 2015;62(3):673-681. |
R835163 (Final) |
Exit Exit |
|
|
Shawlot W, Vazquez-Chantada M, Wallingford JB, Finnell RH. Rfx2 is required for spermatogenesis in the mouse. Genesis 2015;53(9):604-611. |
R835163 (Final) |
Exit |
|
|
Shekh-Ahmad T, Mawasi H, McDonough JH, Finnell RH, Wlodarczyk BJ, Yavin E, Bialer M. Enantioselective pharmacodynamic and pharmacokinetic analysis of two chiral CNS-active carbamate derivatives of valproic acid. Epilepsia 2014;55(12):1944-1952. |
R835163 (Final) |
Exit Exit Exit |
|
|
Shekh-Ahmad T, Hen N, Yagen B, McDonough JH, Finnell RH, Wlodarczyk BJ, Bialer M. Stereoselective anticonvulsant and pharmacokinetic analysis of valnoctamide, a CNS-active derivative of valproic acid with low teratogenic potential. Epilepsia 2014;55(2):353-361. |
R835163 (Final) |
Exit Exit Exit |
|
|
Tabler JM, Barrell WB, Szabo-Rogers HL, Healy C, Yeung Y, Perdiguero EG, Schulz C, Yannakoudakis BZ, Mesbahi A, Wlodarczyk B, Geissmann F, Finnell RH, Wallingford JB, Liu KJ. Fuz mutant mice reveal shared mechanisms between ciliopathies and FGF-related syndromes. Developmental Cell 2013;25(6):623-635. |
R835163 (Final) |
Exit Exit Exit |
|
|
Tsurubuchi T, Allender EV, Siddiqui MR, Shim KW, Ichi S, Boshnjaku V, Mania-Farnell B, Xi G, Finnell RH, McLone DG, Tomita T, Mayanil CS. A critical role of noggin in developing folate-nonresponsive NTD in Fkbp8-/- embryos. Child's Nervous System 2014;30(8):1343-1353. |
R835163 (Final) |
Exit Exit |
|
|
Wallingford JB, Niswander LA, Shaw GM, Finnell RH. The continuing challenge of understanding, preventing, and treating neural tube defects. Science 2013;339(6123):1222002 (7 pp.). |
R835163 (Final) |
Exit Exit |
|
|
Wang B, Jin L, Ren A, Yuan Y, Liu J, Li Z, Zhang L, Yi D, Wang LL, Zhang Y, Wang X, Tao S, Finnell RH. Levels of polycyclic aromatic hydrocarbons in maternal serum and risk of neural tube defects in offspring. Environmental Science & Technology 2015;49(1):588-596. |
R835163 (Final) |
Exit Exit |
|
|
Wang B, Jin L, Ren A, Yuan Y, Liu J, Li Z, Zhang L, Yi D, Wang LL, Zhang Y, Wang X, Tao S, Finnell RH. Correction to levels of polycyclic aromatic hydrocarbons in maternal serum and risk of neural tube defects in offspring. Environmental Science & Technology 2015;49(17):10754-10755. |
R835163 (Final) |
Exit Exit Exit |
|
|
Wang X, Cabrera RM, Li Y, Miller DS, Finnell RH. Functional regulation of P-glycoprotein at the blood-brain barrier in proton-coupled folate transporter (PCFT) mutant mice. FASEB Journal 2013;27(3):1167-1175. |
R835163 (Final) |
Exit |
|
|
Wlodarczyk BJ, Zhu H, Finnell RH. Mthfr gene ablation enhances susceptibility to arsenic prenatal toxicity. Toxicology and Applied Pharmacology 2014;275(1):22-27. |
R835163 (Final) |
Exit Exit Exit |
|
|
Wlodarczyk B, Spiegelstein O, Hill D, Le XC, Finnell RH. Arsenic urinary speciation in Mthfr deficient mice injected with sodium arsenate.Toxicology Letters 2012;215(3):214-218. |
R835163 (Final) |
Exit Exit Exit |
|
|
Yi D, Yuan Y, Jin L, Zhou G, Zhu H, Finnell RH, Ren A. Levels of PAH-DNA adducts in cord blood and cord tissue and the risk of fetal neural tube defects in a Chinese population. Neurotoxicology 2015;46:73-78. |
R835163 (Final) |
Exit Exit Exit |
|
|
Yuan Y, Jin L, Wang L, Li Z, Zhang L, Zhu H, Finnell RH, Zhou G, Ren A. Levels of PAH-DNA adducts in placental tissue and the risk of fetal neural tube defects in a Chinese population. Reproductive Toxicology 2013;37:70-75. |
R835163 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Risk assessment, dose-response, teratogen, organism, cellular, infants, chemicals, toxics, protection, pollution prevention, public policy, environmental chemistry;Relevant Websites:
Finnell Lab | Dell Pediatric Research Institute | The University of Texas, Austin ExitProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.